Services
Services
Production of DNA constructs
We have a 25+ years of experience in recombinant techniques. We witnessed and mastered the classic restriction enzyme based cloning techniques and PCR-based methods, followed by the current waves like Gibson-assembly. We love cloning! If you are interested in more details please contact us at info(at)creativelab.hu address.
Protein production and purification from E. coli
E. coli is the most widely used expression host for heterologous protein expression. It is the best characterized organism and there are ample tools available for manipulation and efficient protein production. High protein yield can be cost-efficiently produced by E. coli. Scale-up is significantly easier than in eukariotic expression hosts, however post-translational modifications are lacking.
High expression levels often lead to insoluble protein producion. Studies show that less than 50 % of bacterial proteins and less than 15 % of non-bacterial proteins can expressed in soluble form. However, a number possible techniques and our experience can improve the outcome, and in vitro refolding is often successful to prepare active proteins. If you are interested in more details please contact us at info(at)creativelab.hu address.

Protein production and purification from CHO cells
Due to its attractive characteristics,
Chinese Hamster Ovary (CHO) cells are biotechnology’s workhorses. First of all, CHO cells are quite easily maintained and can grow to very high densities in suspension cultures and in large bioreactors. They produce correctly folded and post-translationally modified (e.g. glycosilated) proteins that show good compatibility and bioactivity in humans. Transgenes can be introduced and amplified efficiently in the CHO genome reaching high copy numbers of the gene of interest. The more than two decades of research enables the production of proteins
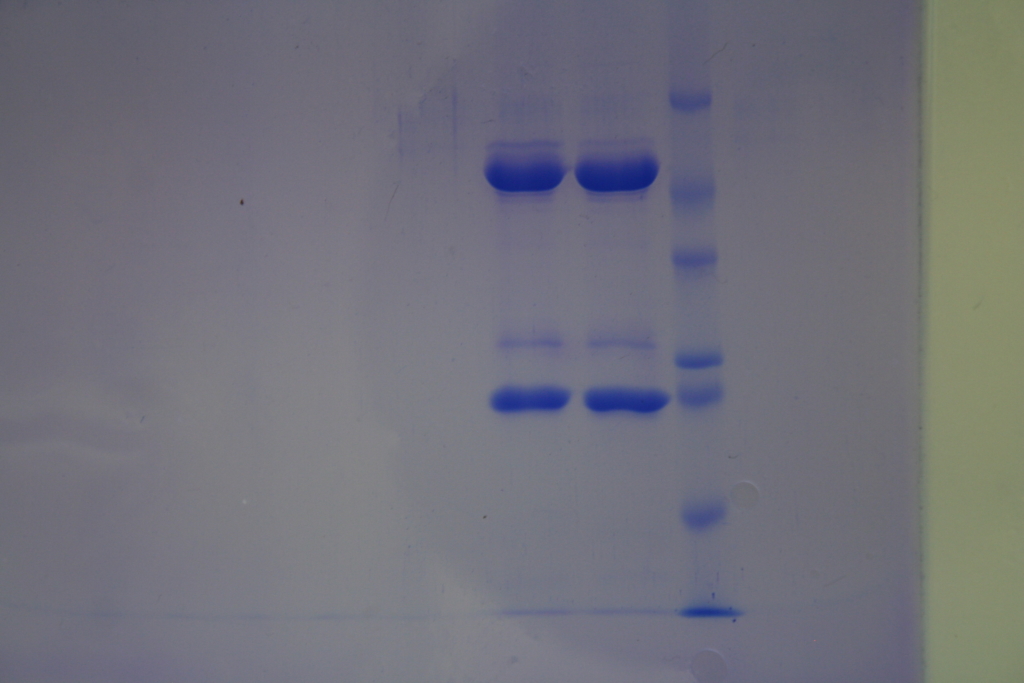
with high yields even up to 5 g/L.
Despite the availability of numerous express
ion hosts, CHO cells have become the most important cell system for the biotechnological of pharmaceuticals. About 70% of all recombinant therapeutic proteins are produced in mammalian cell lines, and CHO is the dominant choice for antibody production. We offer a stable cell line production service in DG44 licensed from Dr. Lawrence Chasin. Our developp
ed technology and kow-how enables the formation of high copy clones with decent protein expression levels. If you are interested in more details please contact us at info(at)creativelab.hu address.




